New digital diagnostic tools should make it possible to better diagnose patients with muscle and joint complaints.
Worldwide, 1.71 billion people experience musculoskeletal complaints. Their journey to the right diagnosis is often long by consulting various practitioners before it is clear what the cause of their problem is.
“We will investigate how doctors deal with the insights from the tool and whether the patient is satisfied. Because that's what it's all about in the end." - Rachel Knevel MD PhD, Project Coordinator
European Cooperation
Working together is key
Within the project, the LUMC is collaborating in a consortium with more than 15 European academic and private partners.
Complex Health Data
Multiple datasources
Electronic Health Record (EHR) data, healthcare registries,
genetics, self reported symptoms, serology and blood work-up are some of the data that will have to be collected and normalized throughout the project.
Privacy & Data security
Data from primary and secondary care
All data from participating countries must be linked to the data scientists in the consortium. IT and privacy departments have set up an appropriate infrastructure that guarantees privacy and data security. Additional information for UK citizens may be found here.
>>>
Approaches
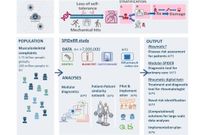
Modular SPIDeRR
A practical support tool for primary care practitioners that provides probabilities for diseases and the usefulness of additional tests and referrals.
Digital twin
A digital twin is the most accurate reflection of reality, whether it be an object, process such as the immune system, organization or person.
Rheumatic
User-friendly questionnaire for patientswith musculoskeletal complaints. Directs patients to the right care and facilitates communication within healthcare.
Overview
A look into the future
SPIDeRR will deliver three clinical models:
✔ A symptom checker for patients
✔ A decision support tool for (primary) care providers, providing guiding additional examination and referral decisions
✔ A patient-patient similarity network to optimize diagnostic groups in rheumatology and support treatment decision.